Application and Review Processes - The ACE Process
Highlights of the ACE accreditation process can be summarized as follows (see remainder of this document for a detailed description of each step):
- Catheterization and PCI facilities initially will be evaluated by a panel of trained nurse reviewers based on written material as required in the ACE Application. The application is designed to verify compliance to all of the standards for any Cath/PCI facility.
- Two independent physician reviewers will review each facility and render an independent opinion. The reviewers will recommend to the Board either “accreditation,” “provisional” until specific improvements have been put in place or volume expectations met, or “deferred”.
- A sample of facilities may be required to undergo an on-site evaluation conducted by physician experts at the expense of the facility. Outlier facilities will be surveyed to validate clinician outcomes. (both high and low performers)
- The panel of reviewers will then render a final recommendation to the ACE Board of Directors for a final decision.
- A written procedure for appeal of a decision will be available.
- Physician reviewers will meet the ACE requirement and be required to attend training session to be qualified to serve as an ACE reviewer.
- Outcome data will be submitted to the accrediting body quarterly, semi-annually, annually or as needed and reviewed to determine substantive changes.
- Corrective action plans will be developed to assist facilities when indicated.
Figure I provides an overview of the ACE accreditation process.
Figure 1
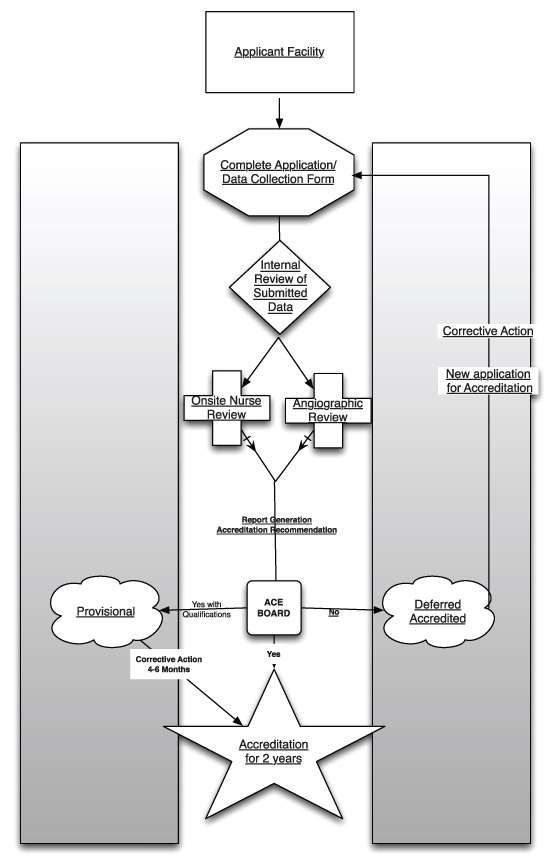 As Figure I illustrates, there are five significant steps to the ACE process. The following sections discuss each in terms of rationale, processes and efforts performed to date.
As Figure I illustrates, there are five significant steps to the ACE process. The following sections discuss each in terms of rationale, processes and efforts performed to date.
Step 1: Applicant Hospitals
There are more than 2,000 cardiac catheterization facilities in the United States. Although there is currently no federal mandate for accreditation of these facilities, several states are drafting policies and legislation that may lead to this occurring. Currently, catheterization laboratory accreditation is a voluntary undertaking.
ACE recognizes that there are several types of facilities that perform these procedures (full service, Emergency PCI without onsite surgery, Elective PCI with-out onsite surgery, diagnostic procedures only, free standing) however the basic requirements for accreditation are similar for all facilities. Specific requirements may be superimposed on these fundamental requirements to address the specific type of facility being reviewed.
One application per facility is accepted and this encompasses catheterization and PCI procedures performed throughout the applicant institution. If multiple facilities within a hospital system perform these procedures, each facility must apply separately for accreditation.
Step 2: Data Collection
Hospitals applying for initial accreditation are required to meet the criteria specified in the ACE Cath/PCI Standards Document. The accreditation process focuses on the equipment, policies, indications, QA processes, patient outcomes and procedures of the institution as they apply to Cath PCI procedures.
As illustrated by Figure 1, both outcomes and process data are collected and reviewed. The key areas of data collected are described in the Standards documents.
Site visits by trained nurse reviewers will validate the reliability of the data. These data and reports are then reviewed by two independent physician reviewers for final recommendation to the ACE Board for final accreditation. Sites where deferred
accreditation is the decision (for cause) may also have site visits performed by physician reviewers. All sites where initial data or site visit reviews have demonstrated sufficient issues to warrant withdrawal or denial of accreditation may have physician site visits.
Step 3: Initial Review
Based upon the required completed application forms and data, the ACE review staff conducts an initial review of each facility applying for accreditation. Following completion of the initial application documents, a site visit is scheduled and performed by trained nurse reviewers.
The ACE review committee provides ongoing support and education to the staff performing these reviews. Independent physician reviewers evaluate all recommendations from the review staff before making a recommendation to the ACE Board of Directors for accreditation.
Step 4: Onsite Review
Following submission of an on-line application and payment of submission fee, ACE will review and notify facility of acceptance for accreditation consideration or ask for additional information or clarification.
ACE staff will then coordinate a one or two day site visit. The ACE staff assigns a team of experienced, trained, objective nurse reviewers to conduct an on site review. The on site review will include but not be limited to the following:
- All Catheterization and PCI laboratories
- Chart audit
- Review of SOP for Cardiac Catheterization/PCI
- Review of QA process and documentation
- Review of operator & staff credentials
- Interview with Medical Director & Key program personnel
- Collection of Angiograms for review by physician panel
- These Angiograms will be randomly selected in advance of the site visit, however additional studies may be requested by the site review team.
- A maximum of 10% of cases per operator will be reviewed including angiography and supporting documentation.
- A minimum of 10 studies per operator will be reviewed.
The nurse reviewers submit a report detailing their findings to the assigned physician reviewers. The physician reviewers are experienced invasive and interventional cardiologists who meet pre- specified criteria and are trained specifically to the ACE standards and tested to verify consistency of evaluation. They review the specific data as well as the summary report from the nurse reviewers. The physician reviewers independently recommend the site for, deferral of accreditation, provisional accreditation or full accreditation to the ACE Board of Directors. Some of the latter facilities may be selected by random for physician site visit. In the case of a disagreement between the physician reviewers, the ACE Board of Directors may either review the application as a whole or assign an additional physician reviewer to assist in consensus building with the initial reviewers (or initiate a physician site visit).
A corrective action plan is developed for facilities not meeting performance standards that include a Provisional period pending repeat review. This period should not exceed 6 months. Depending on the specific deficiencies identified, additional training can be recommended by enrollment in an appropriate training program or more frequent data submission required.
Step 5: Reporting Mechanisms
Outcomes Data (as defined by ACE Data Elements Committee) will be reported in a timely manner in order to maintain accreditation
Participation in a national registry is strongly encouraged but not mandatory as long as all required data is submitted at appropriate intervals. As detailed in both the Standards document for Cath/PCI if the facility is not participating in a national registry, required data must be submitted using comparable definitions to those used by ACC NCDR CathPCI Registry. If the site is not participating in a national registry with standardized definitions, the site visit process will include validation of the data submitted against the nationally excepted, harmonized definitions for the individual data elements. Significant discrepancies between reported data and source documentation review may be grounds for deferral of accreditation.
Major program changes should be reported to ACE these include but are not limited to the following at least yearly:
- Change of the Medical Director
- Major changes to equipment or procedures performed
- Addition/deletion of operators
- Sentinel event as defined by the Joint Commission
- Exceptional occurrences that the facility anticipates affecting accreditation status,
Each facility must complete an annual on-line survey to maintain accreditation.
Facilities with “provisional” status may be asked to submit outcomes data quarterly during the provisional period. They may also be asked to submit additional cases for review as part of the corrective action plan. All facilities, irrespective of their accreditation status may be randomly reviewed without prior notification. These reviews may focus on but are not limited appropriate use assessments, QA processes, and complications.
In developing an approach for conducting this analysis and rendering fair, impartial decisions, multiple documents were utilized. These include the most recent guidelines for catheterization laboratories, PCI performance, quality assurance and appropriate use criteria for revascularization. Additional support for the criteria arise from documents currently under development and expected to be published within the next year as well as the current peer reviewed pertinent medical literature. Outcomes standards were developed based on data available from the ACC NCDR CathPCI and Action registries.