Application and Review Processes - The ACE Process
Highlights of the ACE accreditation process can be summarized as follows (see remainder of this document for a detailed description of each step):
- Carotid stenting facilities initially will be evaluated by a panel of trained reviewers based on written material as required in the ACE Application. The application is designed to verify compliance to all of the standards for any carotid stenting facility.
- Two independent reviewers will review each application and render an independent opinion. The reviewers will recommend to the Board either “accreditation,” “provisional” until specific improvements have been put in place or volume expectations met, or “denial”.
- A sample of facilities will be required to undergo an onsite evaluation conducted by physician experts at the expense of the facility. Outlier facilities will be surveyed to validate clinician outcomes. ( both high and low performers)
- The panel of reviewers will then render a final decision.
- A written procedure for appeal of a decision will be available.
- Outcome data will be submitted to the accrediting body quarterly, semi-annually, annually or as needed and reviewed to determine substantive changes.
- Corrective action plans will be developed to assist facilities when indicated
Exhibit 1 provides an overview of the ACE accreditation process.
As the exhibit 1 illustrates, there are fi ve significant steps to the ACE process. The following sections discuss each in terms of rationale, processes and efforts performed to date.
Exhibit 1
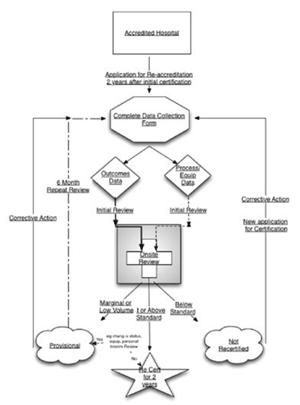
Step 1: Accredited Hospitals
Step #1 in the ACE process (exhibit 3) is that of determining which facilities are eligible for accreditation and re- accreditation. More than 1000 facilities have self-certified to perform carotid artery stenting on CMS beneficiaries. Currently, self- accreditation per the CMS procedures is thesole means to provide accreditation of facilities for carotid stenting. The CMS limited coverage decision for carotid artery stenting, requires all facilities to collect data on carotid stenting at their institutions. In addition, these self-accredited facilities are required to report these data to CMS biannually if requested.
Exhibit 2
At present, it is likely that most facilities that offer (or intend to offer) carotid stenting in the nearterm have already registered with CMS through this self- accreditation process. It is unlikely that the number of facilities will significantly grow so long as the current Medicare coverage restrictions (only high risk, symptomatic patients with > 70% stenosis) remain in effect.
This proposal is based on current realities and addresses several types of facilities, each having unique situations and requirements, it would however be expanded to meet the needs of new accreditations should the opportunity arise:
- For facilities applying for initial accreditation (i.e., having not self-certified under the CMS procedures), little or no consistent performance or outcomes data is generally available. As a consequence, the ACE application and data reporting requirements are focused on the processes in place, equipment, and credentialing of operators. Outcome data will not be available for the facility. To address this, sites may be granted provisional accreditation based on the processes, equipment, and credentialing criteria however, sites will be required to submit clinical data on a quarterly basis until such time as sufficient outcome data are available to adequately assess the results.
- For facilities applying for re-accreditation via ACE (having previously self-accredited under the CMS procedures), two subcategories of facilities are encountered:
- Facilities that have had their self-accreditation for more than 2yrs but have relatively limited experience because of the volume of symptomatic patients treated; these facilities represent the largest group.
- Facilities with substantial experience with carotid stenting extending over a number of years (3-5 years or more) or those with high volumes.
Differences in emphasis are necessary for each of these groups, as described below however both are held to the same outcome standards as described below. On a yearly basis, all facilities will be asked to respond to a brief questionnaire to determine if there have been significant changes in process, equipment or operators. If it is determined that a significant change has occurred, this will prompt more frequent surveillance of outcome data to insure consistency with the previous accreditation.
Accreditation applications are accepted from facilities where cardiologists, radiologists, vascular surgeons and others practice. One application per facility is accepted and this encompasses carotid stenting procedures performed throughout the applicant institution.
Step 2: Data Collection
Applicants for initial accreditation. Hospitals applying for initial accreditation are required to meet the criteria specified in the national coverage decision document as well as standards established by the ACE Carotid Standards Committee. The accreditation process at such facilities focuses on the equipment, policies and procedures of the institution as they apply to carotid artery stenting and compliance with CMS guidelines. Little or no data is generally available for these institutions and outcome data will therefore be reviewed on an ongoing basis at quarterly intervals during a period of provisional accreditation, although experience of the operators applying for credentials are reviewed to determine if they meet subspecialty guidelines for training and competency.
As illustrated by the exhibit above, both outcomes and process data are collected. In the case of initial accreditation, the facility is required to complete a data sheet focusing on the process requirements and submit it for review. The key areas of data collected are described in the Standards and Data document.
Exhibit 3
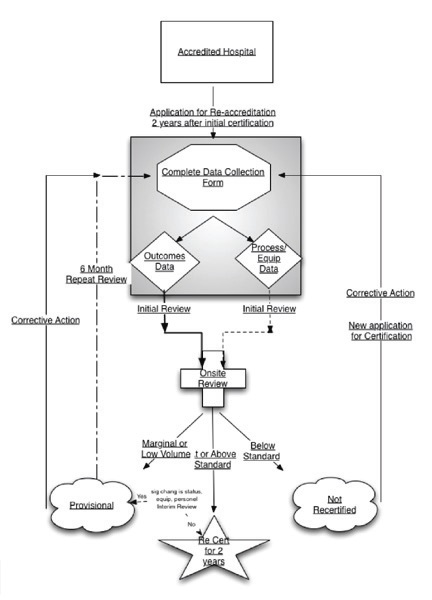
In many institutions carotid stent procedures are performed in a variety of locations (cath lab, radiology suite, operating room). Therefore for each location, the equipment, policies and procedures (if site specific) and system/ operational differences are reported for review.
The majority of facilities obtained their initial accreditation without onsite review based on initial CMS accreditation. Site visits by a trained nurse reviewer will validate the reliability of the data. These data and reports are then reviewed by two independent physician reviewers for final recommendation to the ACE Board for final accreditation. Sites where Provisional accreditationis anticipated (for cause) as well as a random sample of other sites will also have site visits performed by physician reviewers. All sites where initial data or site visit reviews have demonstrated sufficient issues to warrant withdrawal of accreditation will have physician site visits.
Application Process for Initial ACE Reaccreditation (limited experience). Facilities with relatively recent carotid programs or with limited experience, which require re-accreditation, have carotid stent outcomes data available within their institutions. To apply for ACE re-accreditation, those institutions must submit such data for review and analysis. As above, facilities with provisional accreditation, low volumes and limited experience will be expected to submit outcome data on a quarterly basis to retain their provisional status until such time as they qualify for full accreditation. These facilities also are expected to meet pre-established quality standards.
Since those facilities were, by definition, initially self-accredited, the reaccreditation process provides an opportunity for independent review of equipment, policies and procedures to confirm the facility’s compliance with CMS guidelines including appropriateness and inclusion criteria. Quality outcomes are the focus of this review process, but verification of self-reported information is important and necessary for validation of data integrity. Because of the relatively low volume of procedures in this group, statistical comparison to the benchmarks can be difficult because of broad confidence intervals and therefore these facilities require more frequent evaluation (quarterly) to be confident that deterioration of outcomes are not occurring.
Application Process for Initial ACE Reaccreditation (substantial experience). Facilities with a substantial carotid stent experience extending over a number of years: this group requires a less detailed review of equipment, policy, and procedures. This is only to the extent that no significant changes have occurred in the interval since prior accreditation. The focus of this review is on quality and outcomes assessment. The standard to be met is that set by the ACE Carotid Standards Committee but reflecting all data available at the time of the review relative to optimal outcomes of carotid artery stenting in the target populations.
Step 3: Initial Review
Based upon the required completed application forms and data, the ACE review staff conducts an initial review of each facility applying for accreditation. Following completion of the initial application documents, a site visit is scheduled and performed by trained nurse reviewers.
The ACE review committee provides ongoing support and education to the staff performing these reviews Independent physician reviewers evaluates all recommendations from the review staff before making a recommendation to the ACE Carotid Accrediting Board for accreditation.
Exhibit 4

Step 4: Onsite Review
Following submission of an on-line application and payment of submission fee, ACE will review and notify facility of acceptance for accreditation or ask for additional information or clarification.
ACE staff will then coordinate a one or two day site visit. The ACE staff assigns a team of experienced, trained, objective nurse reviewers to conduct an on site review. The on site review will include but not be limited to the following:
- All CAS site facilities
- Chart audit
- Review of SOP for CAS
- Review of operator & staff credentials
- Interview with Medical Director & Key program personnel
- Collection of Angiograms for review by physician panel
Exhibit 5

These reviewers submit a report detailing their findings to the assigned physician reviewers. These individuals are selected from a multidisciplinary pool of experienced carotid stent, vascular medicine and neurologic clinicians who review the specific data as well as the summary report from the nurse reviewers. They independently recommend the site for selected (for cause) physician site visit or accreditation to the ACE Carotid Accreditation board. Some of the latter facilities may be selected by random for physician site visit. In the case of a disagreement between the physician reviewers, the ACE Carotid Accrediting Board may either review the application as a whole or assign an additional physician reviewer to assist in consensus building with the initial reviewers. (or initiate a physician site visit)
A corrective action plan is developed for facilities not meeting performance standardsthat include a Provisional period pending repeat review. Depending on the specific problems, additional training can be recommended by enrollment in an appropriate training program or more frequent data submission required. Based on the corrective plan provided to a particular institution, ACE may recommend to CMS that the institution continue to perform CAS under certain circumstances or that the program be suspended until remediation as recommended is achieved and approved by ACE.
Step 5: Reporting mechnisms
Outcomes Data ( as defined by ACE Data Elements Committee) will be reported in a timely manner in order to maintain accreditation.
Participation in a national registry is encouraged but not mandatory as long as all required data is submitted at appropriate intervals. If the site is not participating in a national registry with standardized definitions, the site visit process will include validation of the data submitted against the nationally excepted, harmonized definitions for the individual data elements.
Major program changes should be reported to ACE these include but are not limited to the following:
- Addition/Deletion of Medical Director
- Major changes to equipment or procedure
- Addition/ deletion of operators
- Sentinal event
- Each facility will complete an annual on-line survey to maintain certification
- Facilities with “provisional” status will submit outcomes data quarterly
In developing an approach for conducting this analysis and rendering fair, impartial decisions, the multispecialty society clinical competency statement for CAS referenced earlier1 provides a useful framework, particularly regarding facilities, equipment and allied personnel for performing CAS:
In addition to the studies that resulted in FDA approval of devices for carotid stenting with distal protection as well as CMS’s national coverage decision, more recent results from post-market approval studies are now available and formed the basis for the development of quality standards. These data allow for more real-time assessment of the characteristics of patients being treated as well as the changing outcomes for the procedure.
In order to provide more valid criteria for reaccreditation, more contemporary outcomes data from clinical trials, post-market surveillance studies, and other registries for the benchmark criteria for outcomes quality assessment will continuously be incorporated as they become available. In light of the current coverage guidelines, it will also be important to evaluate appropriateness criteria for case selection and to confirm (by the on-site review process described above) the documentation regarding patient inclusion criteria in order appropriately evaluate the outcome data.
Exhibit 6

References
- Rosenfield K, Cowley MJ, Jaff MR, Ouriel K, Gray W, Cates CU, Feldman T, Babb JD, Gallagher A, Green R, Kent KC, Roubin GS, Weiner BH, White CW. SCAI/SVMB/SVS clinical competence statement on carotid stenting: training and credentialing for carotid stenting - multispecialty consensus recommendations, a report of the SCAI/SVMB/SVS writing committee to develop a clinical competence statement on carotid interventions. Catheter Cardiovasc Interv, 2005. 64(1):1-11.